| Table of Contents |  |
|
Editorial
| ||||||
| Vitiligo, an ancient but still enigmatic disease | ||||||
| Mohamed Badawy Abdel-Naser | ||||||
|
Prof. of Dermatology, Venereology and STDs, Faculty of Medicine, Ain Shams Uni. Cairo, Egypt.
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] |
| How to cite this article |
| Abdel-Naser MB. Vitiligo, an ancient but still enigmatic disease. Edorium J Dermatol 2015;1:1–4. |
|
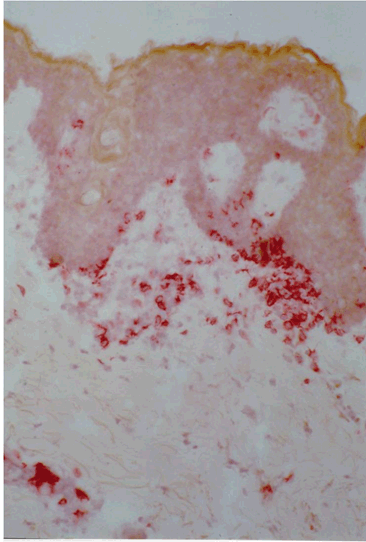
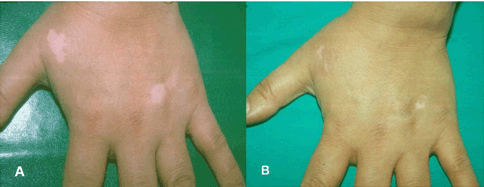
Vitiligo is a common acquired depigmentary disorder that is easy to diagnose but difficult to treat. In this disease, functionally epidermal and occasionally hair bulb melanocytes are selectively targeted and lost resulting in milky white patches with or without leukotrichia. The extent and distribution of vitiligo often change during the course of a person's lifetime and its progression is unpredictable. The disease is simply classified into two main types; namely non segmental (generalized) vitiligo, the common symmetrical form, and segmental vitiligo, affecting only one side of the body (Figure 1A). There is sufficient evidence that the two types not only differ in the pathogenesis of melanocyte loss but also in their response to the different treatment modalities. There are three major hypotheses for the pathogenesis of vitiligo that are not exclusive of each other: biochemical/cytotoxic, neural and autoimmune. Genetic predisposition has been also incriminated in the generalized type based on familial findings but genetic studies are inconsistent [1] [2]. Furthermore generalized vitiligo may be associated with several other autoimmune diseases [3] [4]. Available data provide strong evidence supporting an autoimmune pathogenesis of vitiligo in the far more common generalized type [5] [6] [7]. As vitiligo can have a major effect on quality of life with stigmatization, avoidance behavior and coping, treatment can be considered and should preferably begin early when the disease is active [8]. Unfortunately, medical treatment is not always successful in repigmenting the viliginous skin and not only patients but also physicians are frequently frustrated with the outcome. In order to alleviate this feeling, it is important to explain the difficulty of the scientific understanding of the pathogenesis of vitiligo. Melanin pigment, which is responsible for skin color, is synthesized by the melanocytes which are of neural crest origin that migrate during embryogenesis to reside mainly in the eyes, the epidermis and hair bulbs. However, melanin must be transferred to adjacent keratinocytes by engulfing (phagocytising) terminal melanocyte dendrites with their melanin contents to give the skin its characteristic color. Failure of melanin transfer will result in white depigmented skin. It has been shown that this process of phagocytosis is exerted by basal and viable suprabasal keratinocytes and melanin is carried to outermost layers by the process of keratinocyte division and migration. Migration time of basal keratinocytes with their melanin contents to the outermost layers is 26–42 days; denoting that patients presenting with established depigmented patches must have their melanocytes lost at least several weeks before this time of presentation [9]. This phenomenon makes examining the early changes of the pathogenesis of vitiligo almost impossible and most findings described in literature are rather representing late findings and after melanocyte destruction and cessation of melanin transfer. Accordingly, the most acceptable studies of vitiligo pathogenesis in humans are those obtained from the border between depigmented and pigmented skin (perilesional skin) and while the disease is still active and spreading. These studies consistently revealed the presence of mixed lymphohistiocytic infiltrate, with predominant cytotoxic T lymphocytes in addition to upregulation of several inflammatory cytokines and adhesion molecules, such as IFN ?, TNFa, and ICAM-1 among others. In vitro studies also revealed evidence of an antibody-dependent cellular cytotoxicity; accordingly it is likely that not only cellular but also humoral immunity is involved in the pathogenesis of vitiligo (Figure 2) [5] [10] [11]. The cellular cytotoxicity mechanism has been confirmed in an in vitro skin explants model in which perilesional T cells from vitiligo patients selectively targeted and destructed epidermal melanocytes [6]. Other factors, such as genetic predisposition, oxidative stress, and auto-cytotoxicity may play a role in triggering this response. Opponents of the autoimmune mechanism cast doubt on it due to the paucity of the infiltrate and the difficulty of demonstrating the anti-melanocyte antibodies. However, they are overlooking two important facts namely, that all in vivo studies revealed late findings as already explained and several weeks after completion of melanocyte destruction and hence down regulation of the auto immunological reactions. The second fact is the fastidious nature of melanocytes as shown in vivo by their easy destruction by cryotherapy, burns and chemicals (substituted phenols) and in vitro by the difficulty of their selective cultivation requiring several melanocyte mitogens and limited life span [12]. In the light of these inherent difficulties in fully understanding the pathogenesis of melanocyte damage and loss in vitiligo, current treatment modalities are directed towards stopping progression of the disease and achieving repigmentation. Corticosteroids (systemic and topically) and other topical immunomodulators are helpful in cases of active vitiligo and vitiligo of recent onset and before complete destruction of melanocytes. The mechanism of action is to inhibit the autoimmune/inflammatory reaction. Photo(chemo)therapy (such as topical/systemic PUVA, narrow band UVB and excimer laser) is instead of are effective in early and active cases and also in well established cases [13] [14] [15] [16]. The mechanism of action is not only to inhibit the autoimmune reaction but also to mainly stimulate division and migration of hair bulb melanocytes and melanocytes from the adjacent pigmented skin; unfortunately division of melanocytes is limited and their migration is inherently slow (Figure 1B). Medical treatments therefore require several months or years in order to achieve favorable results and this delayed and slow repigmentation response is an additional reason for patient and physician frustration. Furthermore acral lesions and lesions over boney prominences are known to be resistant to all medical treatments as they lack hairs which are the main source of replicating and migrating replenishing melanocytes. To circumvent these limitations, surgery, such as split thickness skin grafts, minigrafting, suction blister grafting and non-cultured and cultured melanocytes can be applied to difficult to treat areas in stable vitiligo cases and after failure of adequate medical treatment [17] [18] [19]. In general, surgical treatment, which is practically a melanocyte transfer, is suitable for localized lesions of generalized vitiligo and cases of segmental vitiligo which is known to be less responsive to medical therapy. The choice of the procedure depends on physician's comfort and experience (Figure 3) (Figure 4). Combination of medical and surgical treatments is usually advisable as it may improve the surgical outcome. A further concern is the criteria of stable vitiligo which is one of the prerequisites of the success of surgery; so far there is no general consensus about them and loss of the transplanted melanocytes may occur, i.e., Koebner phenomenon [20] [21]. It is at this point that the frustration reaches its maximum and patients lose hope of cure and they are not willing to try any further treatments. Cosmetic options provide ultimate help and include makeup, a self-tanner and skin dye [22]. In cases of widespread vitiligo usually involving large areas (>80% of the entire integument) depigmentation of normally pigmented skin seems plausible [23]. In conclusion, the frustration of patients and physicians alike from vitiligo pathogenesis and hence treatment is understandable and expected, nevertheless further research of this disorder and the advances of cell and tissue culture techniques will greatly and continuously improve our understanding and treatment of this enigmatic ancient disease. |
|
|
|
|
|
|
|
|
|
|
|
References
|
|
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions:
Mohamed Badawy Abdel-Naser – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2015 Mohamed Badawy Abdel-Naser. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|